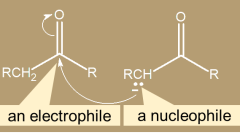
Michael reaction
1,4-addition is called conjugate addition. Using enolate ions as nucleophiles in conjugate addition, this is the Michael reaction.
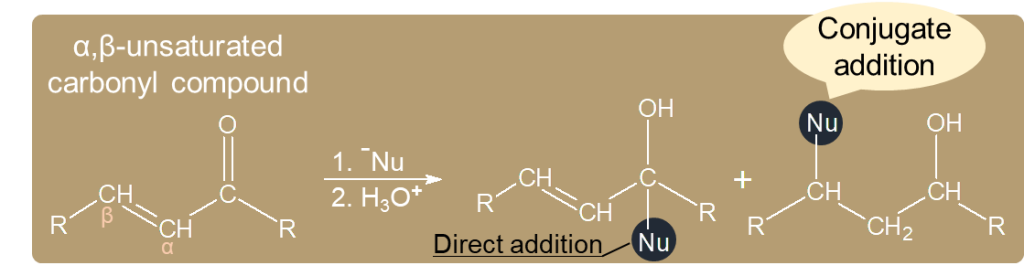
- The reaction is addition of an α-Carbon to the β-Carbon of an α,β-unsaturated carbonyl compound.
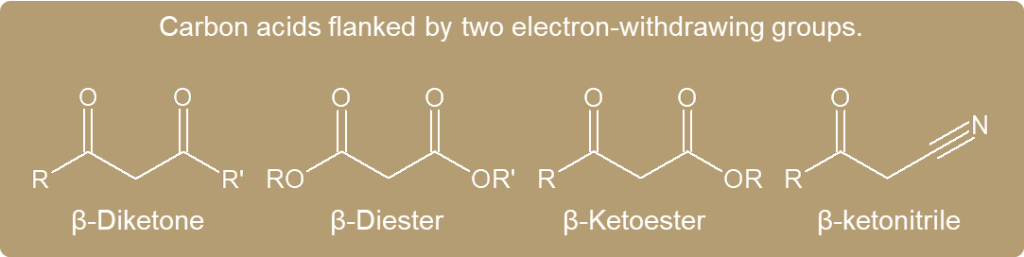
- The enolate ions that work best are those formed from carbon acids that are flanked by two electron-withdrawing groups.

The base used to remove the α-proton must be the same as the leaving group of the ester.
Stork reaction -Michael reaction using Enamines-

Enamines react with electrophiles in the same way that enolate ions do.
- Formation of the enamine. click here
- This enamine undergoes Michael addition to 3-buten-2-one, which after hydrolysis produces a 2-(3-Oxobutyl)cyclohexan-1-one (1,5-diketone).