 8-19
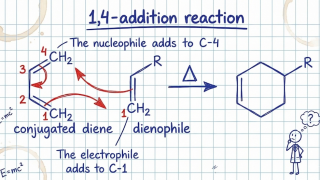
8-19 Diels-Alder Reaction
The Diels–Alder reaction is a concerted reaction: the addition of the electrophile and the nucleophile occurs in a singl...
 8-19
8-19  18-13
18-13  6-11
6-11  18-11
18-11 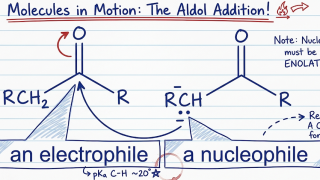 18-10
18-10 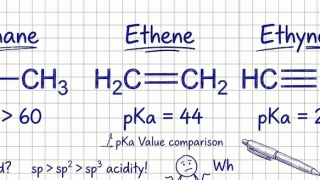 7-11
7-11 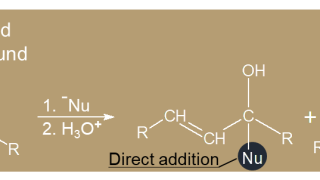 18. α-Carbon
18. α-Carbon 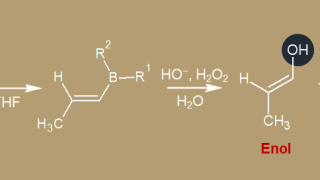 07. Alkynes
07. Alkynes 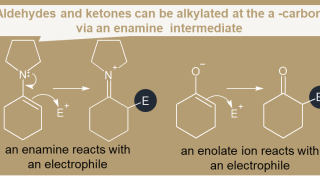 18. α-Carbon
18. α-Carbon  06. Alkenes
06. Alkenes  6-11
6-11  06. Alkenes
06. Alkenes  7-11
7-11  07. Alkynes
07. Alkynes