Ozonolysis of alkenes
When an alkene is treated with ozone (O3) at a low temperature, the double bond break and the carbons that were doubly bonded to each other are now doubly bonded to oxygens instead.

Ozonolysis is an oxidation reaction because the number of C—O bonds increases.
Ozonide formation
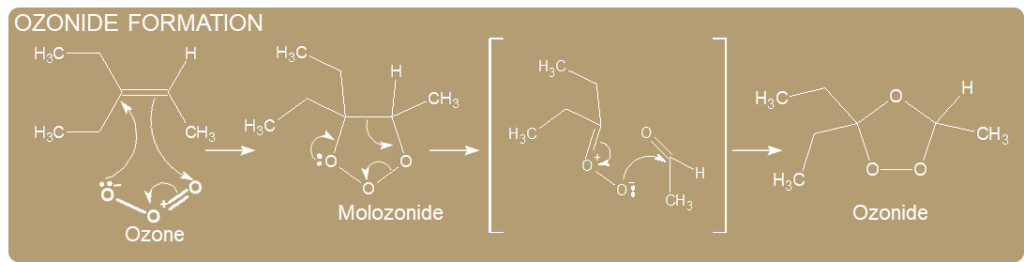
The molozonide is unstable because it has two O-O bonds; it immediately rearranges to a more stable ozonide.
Ozonolysis
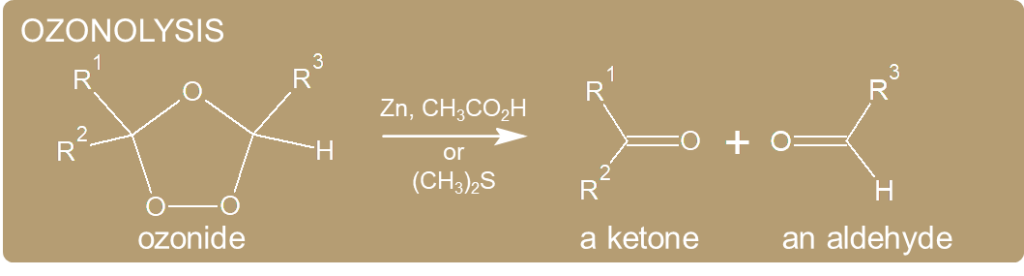
- Ozonide is highly explosive and is usually decomposed using dimethyl sulfide. Dimethyl sulfide attacks ozonide and turns itself into DMSO.
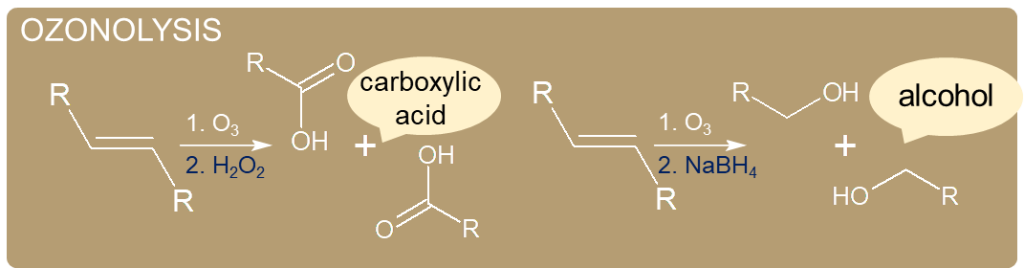
- Ozonide also reacts with oxidants such as H2O2 to form carboxylic acids.
- On the other hand, a strong reducing agent such as NaBH4 will turn it into an alcohol.